The FDA announcement about the Advisory Committee meeting on the Pfizer COVID-19 vaccine booster for September 17, 2021 is here. The time will be 8:30 AM-3:45 PM ET (check your time zone provided on the FDA YouTube web page). Review materials (if available) will be posted at the FDA link provided in the announcement.
Category: Brief Messages or Announcements
Comirnaty vs Comiranty Spelling Bee Issue Resolved

This is just an update on the Pfizer-BioNTech COVID-19 vaccine Comirnaty vs Comiranty spelling issue at UIHC, which has been resolved today. It took a few emails to get it fixed. I was beginning to think I was losing my mind and that I was the only who could see the mistake. Comirnaty had apparently been misspelled as Comiranty on several web pages for maybe a couple of weeks.
You can just look at the word Comirnaty and see how this could have happened. Looking at it in print makes me think there are two letter “m’s” in it. Transposing the two letters “a” and “n” looks easy to do. There are a few anagrams web sites that are picking up on the word Comirnaty. I kind of like “try anomic.” Can you really get “community,” “immunity,” and “mRNA” out of that agglomeration? Maybe. It’s a name game.
I could chalk this up to just me being a retired guy with too much time on his hands and nitpicking. On the other hand, there is that story about a typo ending World War II. In all fairness, there is some doubt about the accuracy of it. But it’s fascinating to think that the difference in spelling between cryptogamist (someone who studies algae) and cryptogramist (someone who studies codebreaking) might have made all the difference in the war’s outcome.
Comirnaty Misspelled by the Medical Community
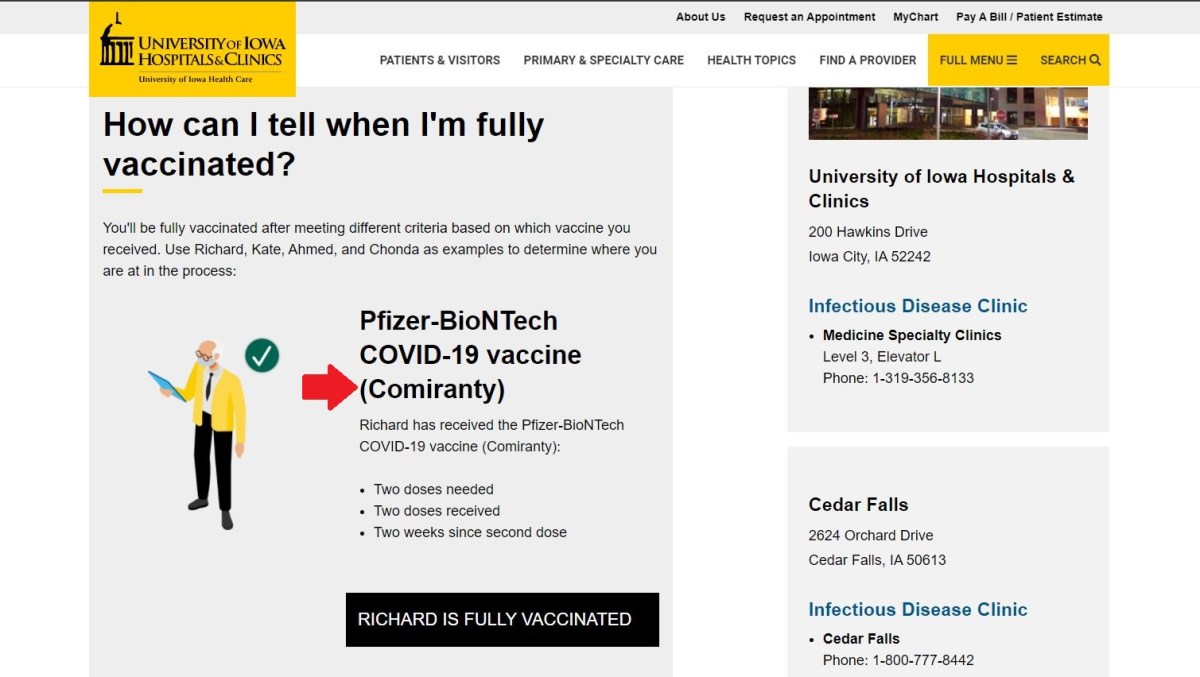
This afternoon I just notified somebody at the University of Iowa Hospitals & Clinics (UIHC) that the Pfizer COVID-19 vaccine’s name is misspelled on several of their web pages. The new name for the vaccine is Comirnaty (pronounced koe-mir-na-tee). UIHC misspells it as “Comiranty.” And it has been that way for at least a week, probably since Pfizer publicly announced the name after the vaccine was fully licensed by the FDA. I found three instances of that although there could be more.
I found a news item that explains the name was deliberately chosen in order to remind us of the word “community” and the “mRNA” technology of the vaccine.
It actually reminded me of Foster Brooks whose comedy routine consisted of acting like he was drunk, slurring his speech in a parody of intoxication. The only way “Comirnaty” can make you think of “community” is if you’ve had a few too many.
On the other hand, “Comiranty” makes me think of the one Indiana Hoosier football player who was caught on camera with the word Indiana misspelled as “Indinia” on his jersey yesterday. By the way, Iowa beat Indinia 34-6 in the season opener. In all fairness, Indiana is not the only state that struggles with spelling.
Update on COVID-19 Vaccine Boosters

The messaging on COVID-19 boosters is being clarified by the CDC and the FDA in separate announcements, posted September 1, 2021.
The CDC update is here. The FDA announcement is here. The FDA plans to hold a virtual meeting about the issue September 17, 2021 from 8:30 AM to 3:45 PM EST and it will be livestreamed on the agency’s YouTube channel. Background material will be made available to the public two days before the meeting.
“Those who are tardy do not get fruit cup.”
The COVID-19 Delta Variant

I’m passing along the University of Iowa Hospitals & Clinics information about the Delta variant of COVID-19. If you’re vaccinated and you get infected with the Delta variant, you might feel like your symptoms are from allergies or a cold. If you’re not vaccinated, you’re a lot more likely to get sick enough to be hospitalized.
According to Dr. Claudia Corwin, MD, MPH, an occupational medicine specialist and associate director of the University Employee Health Clinic, about 97% of those with a severe case of Delta variant are unvaccinated. That doesn’t mean the vaccines make us bullet proof and breakthrough infections of the Delta variant occur.
That’s probably part of the reason why the Advisory Committee on Immunization Practices (ACIP) thinks vaccine booster shots might be better reserved for the most vulnerable patients, such as the very elderly, those in Long Term Care Facilities, and health professionals. The ACIP met yesterday and there was no firm decision about booster rollout specifics and there was no vote on the matter. They plan to meet again in mid-September to review the need for boosters.

Advisory Committee On Immunization Practices Meeting On Boosters Today

Judging from the slides in Dr. Sarah Oliver’s presentation, “Framework for booster doses of COVID-19 vaccines” in the ACIP meeting this afternoon, there is limited data to support COVID-19 vaccine boosters for the general population at this time. The target populations for boosters would be the residents of Long Term Care Facilities (LTCF), health professionals, and those over 65 and 75 years of age, although the goal of ensuring that as many unvaccinated individuals get vaccinated should be actively pursued. There will continue to be further meetings to discuss the role of boosters.
FDA Approves First COVID-19 Vaccine

Today the FDA approved the Pfizer COVID-19 vaccine (now marketed as Comirnaty). Excerpt of the media announcement below:
“The FDA’s approval of this vaccine is a milestone as we continue to battle the COVID-19 pandemic. While this and other vaccines have met the FDA’s rigorous, scientific standards for emergency use authorization, as the first FDA-approved COVID-19 vaccine, the public can be very confident that this vaccine meets the high standards for safety, effectiveness, and manufacturing quality the FDA requires of an approved product,” said Acting FDA Commissioner Janet Woodcock, M.D. “While millions of people have already safely received COVID-19 vaccines, we recognize that for some, the FDA approval of a vaccine may now instill additional confidence to get vaccinated. Today’s milestone puts us one step closer to altering the course of this pandemic in the U.S.”
COVID-19 Boosters for The Immunocompromised: University of Iowa Hospitals & Clinics

Information from The University of Iowa Hospitals & Clinics on COVID-19 vaccine boosters for the immunocompromised is here.
Advisory Committee on Immunization Practices Meeting on Boosters

There is a meeting of the Advisory Committee on Immunization Practices (ACIP) scheduled for August 30, 2021 to discuss COVID-19 vaccine boosters. It looks like it was originally scheduled for August 24, 2021 but was rescheduled. This link to the Federal Register announcement identifies the matters to be considered (booster doses) of the meeting although the date for the meeting had not been updated at the time I wrote this post. Further ACIP meeting information is here.
COVID-19 Vaccine Booster Doses May Be available September: Joint Statement from HHS PUblic Health & Medical Experts

The CDC and FDA released a Joint Statement today indicating COVID-19 vaccine booster doses for all Americans possibly beginning as soon as September 2021. An excerpt is below:
“We have developed a plan to begin offering these booster shots this fall subject to FDA conducting an independent evaluation and determination of the safety and effectiveness of a third dose of the Pfizer and Moderna mRNA vaccines and CDC’s Advisory Committee on Immunization Practices (ACIP) issuing booster dose recommendations based on a thorough review of the evidence. We are prepared to offer booster shots for all Americans beginning the week of September 20 and starting 8 months after an individual’s second dose. At that time, the individuals who were fully vaccinated earliest in the vaccination rollout, including many health care providers, nursing home residents, and other seniors, will likely be eligible for a booster. We would also begin efforts to deliver booster shots directly to residents of long-term care facilities at that time, given the distribution of vaccines to this population early in the vaccine rollout and the continued increased risk that COVID-19 poses to them.”